


End-Capping π-Conjugated Systems with Medium-Sized Sulfur-Containing Rings: A Route Towards Solution-Processable Air-Stable Semiconductors
Aiko Fukazawa, Yusuke Toda, Masahiro Hayakawa, Anna Sekioka, Hiroyuki Ishii, Toshihiro Okamoto, Jun Takeya, Yuh Hijikata, Shigehiro Yamaguchi, Chem. Eur. J., Early View.
[DOI: 10.1002/chem.201802656]
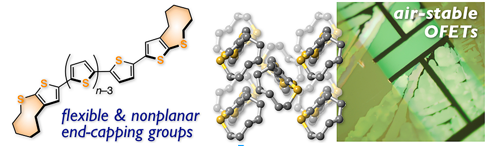
The sulfur‐containing nine‐membered heterocycle thiacyclononene (TN) was evaluated as a new type of end‐capping group for π‐conjugated systems. A systematic study on TN‐capped α‐oligothiophenes (TNnTs; n=4–7) revealed that the capping with TN, which adopts a bent conformation, imparts the resulting oligothiophenes with drastically increased solubility at approximately 140 °C and high electrochemical stability, whereas the electronic structure remains virtually unperturbed. The even‐numbered oligothiophenes TN4T and TN6T form characteristic offset herringbone‐type packing structures on account of the steric repulsion between the TN rings and the presence of intermolecular nonbonding S⋅⋅⋅S interactions. This packing mode in combination with the high solubility enabled the solution‐process fabrication of field‐effect transistors based on TN6T, which exhibited a high performance without degradation even upon exposure to air.
硫黄を含む中員環でエンドキャップする:大気安定な塗布型有機半導体のための新たな置換基のデザイン
2018/08/02
